On February 10, 2021, the Commission accepted commitments offered by South African pharmaceutical company Aspen and ended one of its rare investigations into excessive pricing (and reportedly the first in the pharmaceutical sector).[1] The decision provides guidance on how the Commission evaluates excessive pricing of off-patent medicines and how to remedy potential concerns.
In 2017, the Commission started an investigation into Aspen’s steep price increases of six off-patent cancer medicines in the previous five years. In its preliminary assessment, the Commission was concerned that Aspen abused its dominant position in several national markets in the EEA by imposing excessive prices. Aspen allegedly succeeded in imposing those prices by threatening to withdraw the medicines—most without alternatives—from the national lists of reimbursable drugs.
The Commission’s methodology in its preliminary assessment was to analyze Aspen’s profitability by looking at its accounting data. The Commission found that Aspen’s prices exceeded its relevant costs by almost 300% on average, even after accounting for a reasonable rate of return. Aspen’s average profitability in the EEA was three times higher than the average profitability of comparable companies. The Commission found no legitimate reasons for these high profit margins. Aspen did not improve the medicines or their distribution, nor would it recoup R&D investments because the medicines were off-patent for 50 years. The Commission’s preliminary assessment therefore found that Aspen likely abused its dominant position by imposing excessive prices.
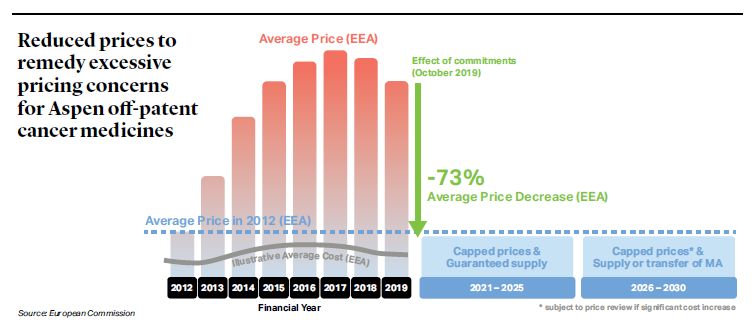
To end the probe, Aspen committed to decrease the prices of the medicines to a fixed price ceiling for the next 10 years in the EEA Member States where the medicines are sold (retroactively taking effect as of October 1, 2019).[2] This entails a price decrease of 73% on average in the EEA (see picture below). Aspen further guarantees to supply the medicines for the next 5 years, and for an additional 5 years will either supply the medicines or make the marketing authorization available to other suppliers. The commitments do not cover Italy because the Italian competition authority took a separate decision against Aspen in 2016.[3]
Commission decisions on excessive pricing are rare, and Commissioner Vestager emphasized in her press statement that the Commission is not a price regulator.[4] She continued to say, however, that the Commission will intervene when a company abuses its dominant position by imposing excessive prices. The decision in Aspen provides welcome guidance on what the Commission considers to be ‘excessive’ and how to remedy abusive price practices in future cases affecting the pharmaceutical sector.
[1] Commission Press Release IP/21/524, “Antitrust: Commission accepts commitments by Aspen to reduce prices for six off-patent cancer medicines by 73% addressing excessive pricing concerns,” February 10, 2021. The text of the Commission’s decision accepting Aspen’s commitments has not been published yet.
[2] Aspen (Case COMP/AT.40394), final commitments of February 10, 2021.
[3] Autorità garante della concorrenza e del mercato, Case A480 Price increase of Aspen’s medicines, decision of September 29, 2016.
[4] Commission Press Release STATEMENT/21/526, “Statement by Executive Vice-President Vestager on the Commission decision to accept commitments by Aspen to reduce prices for six off-patent cancer medicines by 73% addressing excessive pricing concerns,” February 10, 2021.